Thomson's dilemma: how could matter containing electrons be neutral and where was all the mass? Other experiments with discharge tubes suggested the existence of a positive particle with much greater mass (the proton). With this value, and the charge/mass ratio that Thomson had measured earlier it was possible to calculate the mass of the electron. His eventual conclusion was that the charges on the drops were either equal to or multiples of one number which he decided was the charge of a single electron. Millikan used his apparatus to make many measurements of the effect of the electric field vs. Thus there are obviously particles smaller than atoms. 
Using the smallest charge obtained and Thomson's charge/mass ratio the electron mass is roughly 1/2000 the mass of the lightest atom. In 1909 Robert Millikan used the classic oil drop experiment to determine the charge on these particles. He was able to measure the charge/mass ratio of these particles and found this to be the same regardless of what gas was in the tube or what metal the electrodes were made from. Thomson demonstrated in 1897 that the rays consist of a stream of negatively charged particles which he called electrons. From careful experiments with cathode ray tubes J.J. Thus these tubes have been called cathode ray tubes. /GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)
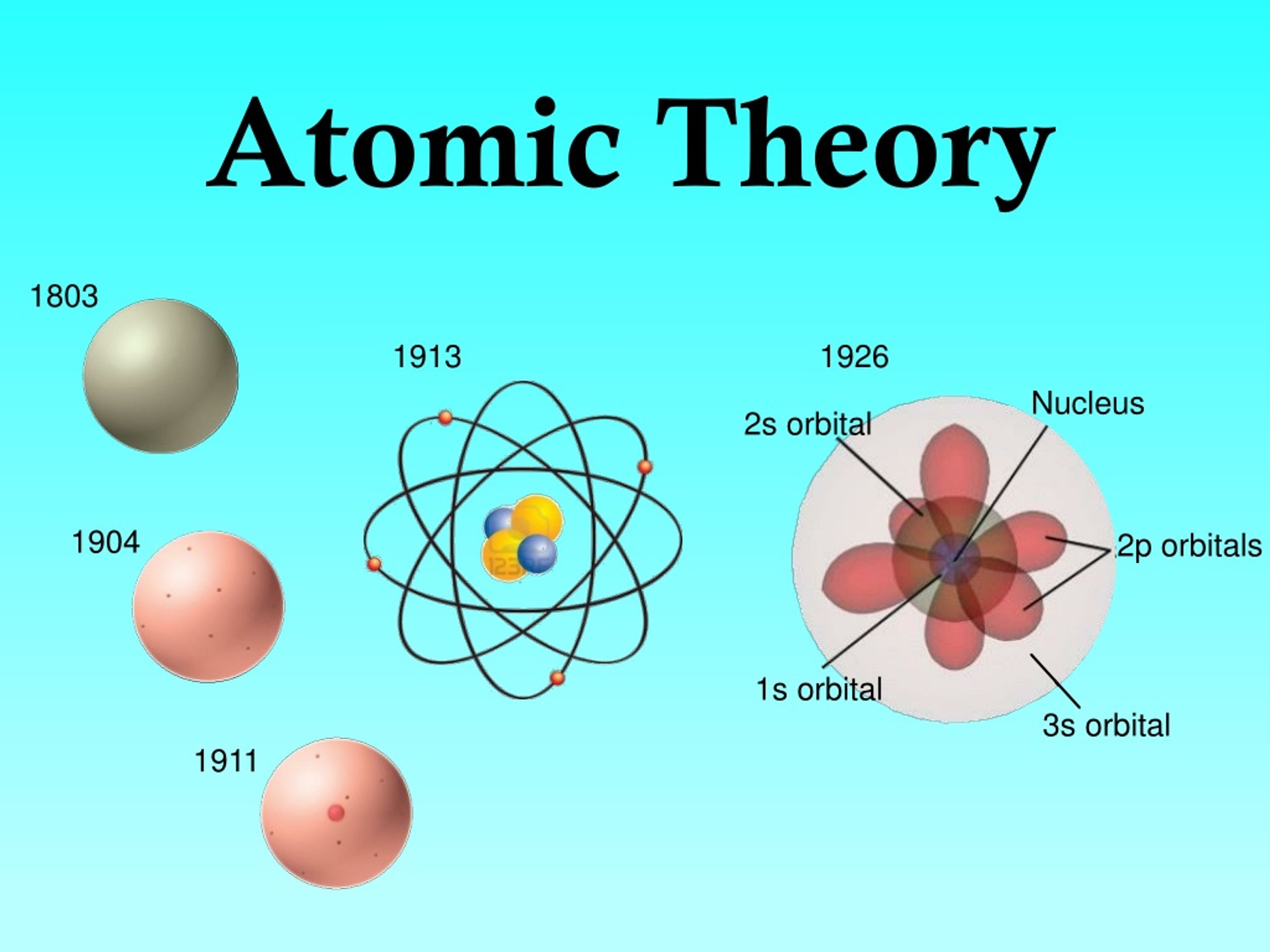
Thomson (seen here in his lab with one of his many hand-made cathode ray tubes)Īssociated with the flow of electricity in such a tube are rays which originate from the negative electrode-the cathode. The first evidence for sub-atomic particles came from experiments with the conduction of electricity through gases in sealed glass tubes at low pressures.įigure : J.J. An eminent organic chemist of the time, Adolf Kolbe, said in 1877, "Dalton's atoms are no more than stupid re table-tapping and supernatural explanations." Some contemporaries of Dalton remained unimpressed. compounds result from combinations of atoms in specific ratios.atoms of an element are identical to one another but different from atoms of other elements.atoms of one element cannot be converted into atoms of another element.all matter consists of indivisible atoms which cannot be created or destroyed.This led to a relative mass scale for atoms that was mostly in error.ĭalton also had his own notation for some of the elements he recognized.ĭalton's "modern" atomic theory, in todays terms, would be something like this: He described what we now call molecules as "atoms" and was convinced that the simplest binary compound of any two elements would have those atoms in a 1:1 ratio. His experiments with gases eventually led him to the idea that matter came in small indivisible units but his use of the word "atom" was variable. John Dalton apparently thought that atoms were pretty much little solid spheres without internal structure. And the third postulate led Dalton to formulate the Law of Multiple Proportions. It may not seem like much, but such a theory was used to explain two of the major laws in chemistry: the Law of Conservation of Mass and the Law of Constant (definite) Composition. In 1808 the first statement of a modern atomic theory was published by John Dalton, a Quaker schoolmaster from Manchester.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
